FDA approves first over-the-counter birth control pill
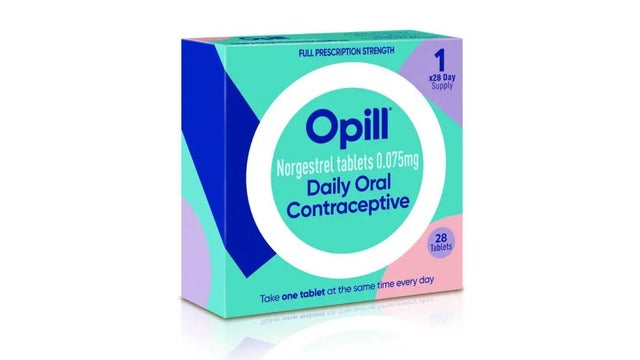
The U.S. Food and Drug Administration has approved the first over-the-counter birth control pill, allowing Americans to buy a daily oral contraceptive without a prescription.
Opill, the progestin-only pill from drugmaker Perrigo, will provide an option for obtaining oral contraceptives without needing to first see a health care provider, in hopes of reducing barriers to access, according to the FDA's news release, which notes that almost half of the 6.1 million pregnancies in the U.S. each year are unintended.
"Today's approval marks the first time a nonprescription daily oral contraceptive will be an available option for millions of people in the United States," Dr. Patrizia Cavazzoni, director of the FDA's Center for Drug Evaluation and Research, said in a news release Thursday morning. "When used as directed, daily oral contraception is safe and is expected to be more effective than currently available nonprescription contraceptive methods in preventing unintended pregnancy."
Opill is made up of norgestrel, a kind of "progestin-only" birth control pill that was first approved as safe and effective to be prescribed by doctors in the 1970s. This is different from birth control pills that are largely prescribed today, which are newer "combined" formulations that also use estrogen.
This is a developing story and will be updated. Alex Tin contributed to this report.
source: https://www.cbsnews.com/news/fda-approves-first-over-the-counter-birth-control-pill-opill/